 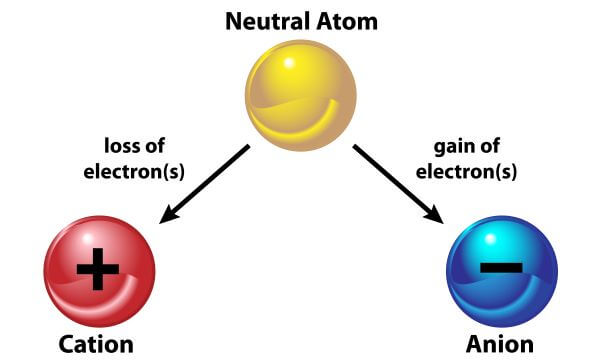
When Ca combines with Cl, reactions 2 and 3 occur, resulting in the molecule CaCl 2. When Na interacts with O, reactions 1 and 4 occur, yielding Na as the resulting chemical. Now, when Na combines with Cl, reactions 1 and 3 occur, yielding NaCl as the resulting product. The elements and the ions generated by them when they lose or gain e are shown in the table below. This is because ions, which act as charge carriers, are present. Ionic bond molecules are strong conductors of electricity in their aqueous solutions or molten form. Molecules that are ionically bound have a high melting and boiling point.Ĥ. Ionic bonds are the most reactive of all the bonds in the suitable medium since they have charge separation.ģ. Ionic bonds are the most powerful of all bonds.Ģ. The following qualities are found in ionic bonded molecules due to the presence of a strong force of attraction between cations and anions:ġ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
